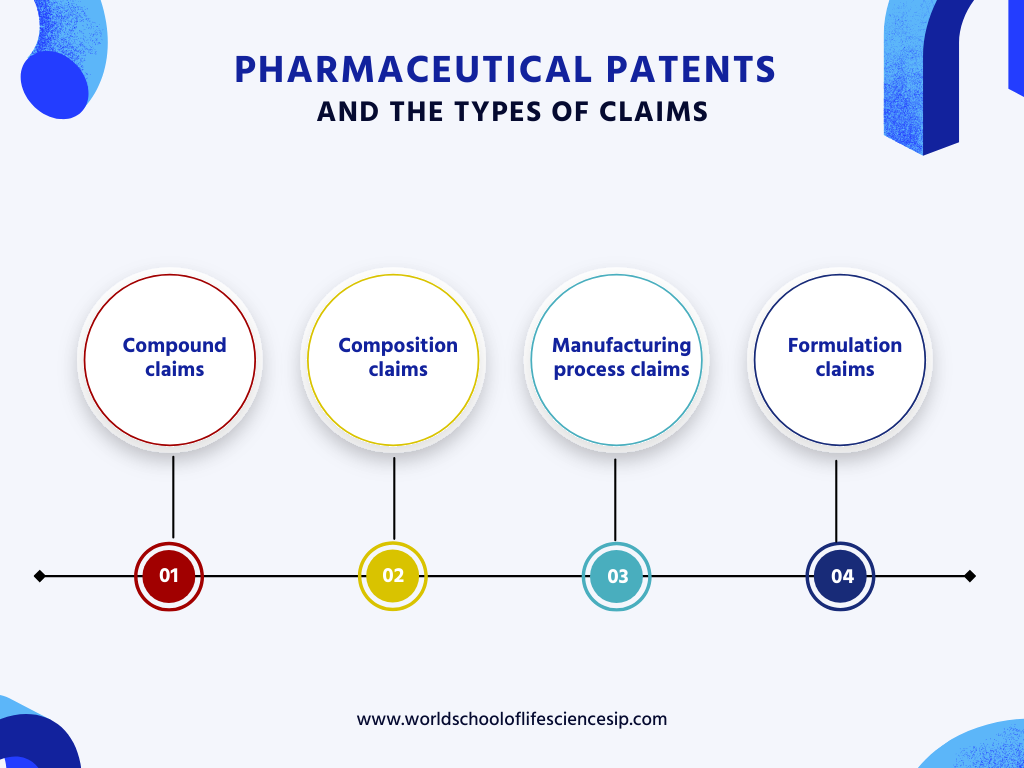
Complex Landscape of Xarelto® in the EU: A Comprehensive Overview
Rivaroxaban, marketed under the brand name Xarelto®, has emerged as a pivotal therapeutic option for various cardiovascular conditions, revolutionizing the landscape of anticoagulant therapy. Its